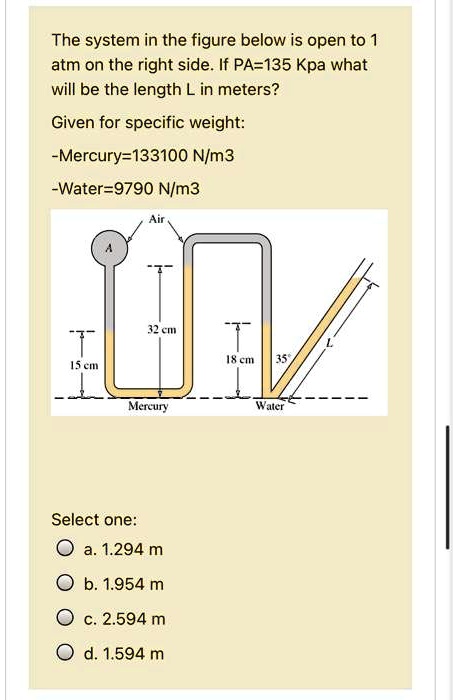
SOLVED: The system in the figure below is open to 1 atm on the right side. If PA = 135 kPa, what will be the length L in meters? Given specific weight: -
The guage pressure exerted below a column of water, open to the earth's atmosphere at depth of 10 m is (density of water = 1000 kg/m3, g = 10 m/s2 and 1 atm pressure = 105 Pa)

Gas Laws Chapter 5. Pressure Force per unit area Measured in Atmospheres ( atm) Mm of Hg = Torr Pascals or kiloPascals (Pa or kPa) - ppt download

How to convert Atmospheric pressure to Pascal (atm-Pa) and Pascal to Atmospheric pressure (Pa-atm). - YouTube

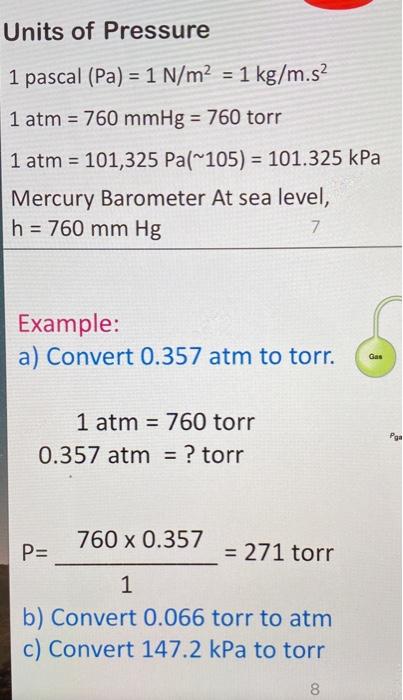
Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download

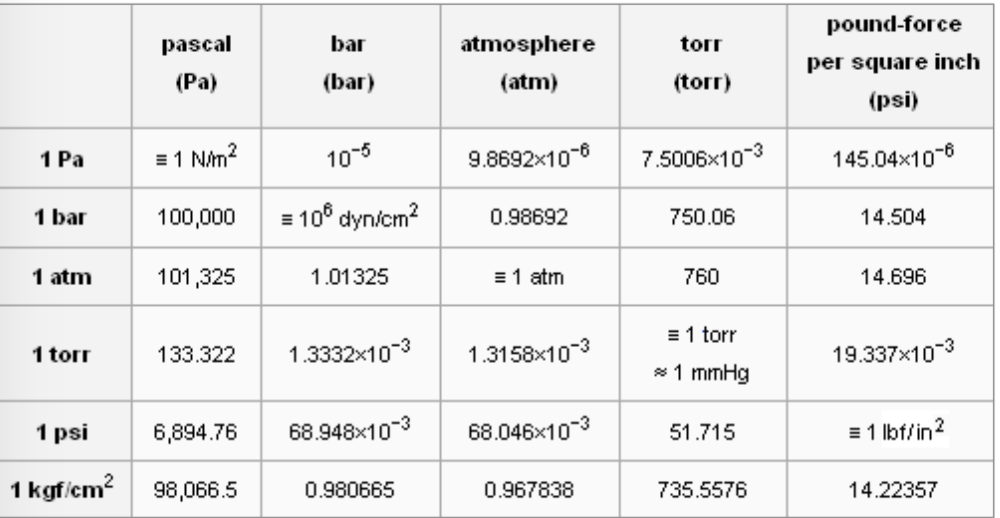
SOLVED: Convert 661,000 pascal, pa into torr: 1atm = 760 torr = 760 mmHg = 101,325 pa 101.325 kpa 1.01325 barr 14.696 p.s.i. 29.921 inHg (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures)

:max_bytes(150000):strip_icc()/GettyImages-124564231-58b5c7d75f9b586046caddb9.jpg)










![Gujrati] 1 atm = ……. Pascal Gujrati] 1 atm = ……. Pascal](https://static.doubtnut.com/ss/web-overlay-thumb/7033101.webp)






